ACIDS,BASES AND SALTS

In our daily life we make use of variety of substances, they are differ in taste and physical state. The substances may be either acids ,bases or salts.
ACIDS
The word acid comes from the latin word acere which means sour .Examples of acids: vinegar, lemon juice,orange juice, curd,tamarind,amla,black tea,grape etc.
CHARACTERISTICS OF ACIDS
- Acids are sour in taste
- they are corrosive in nature
- they are good conductors of electricity
- as they allow the passage of current through them.
- they turn blue litmus to red.
TYPES OF ACIDS

MINERAL ACIDS:-these are acids prepared from minerals present in the earths crust.
ORGANIC ACIDS:-these are acids produced by plants and animals .
WEAK ACIDS:-these acids do not completely dissociate in solution.
STRONG ACID:-these dissociate completely in solution.
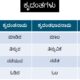
| NAME OF THE ACIDS | FOUND IN |
| Lactic acid | curd |
| Oxalic acid | spinach |
| Ascorbic acid | amla |
| Citric acid | lemons |
| Tartaric acid | tamarind |
| Formic acid | Ants sting |
| Carbonic acid | sodas |
CHARACTERISTICS OF BASES
- There are bitter to taste
- Soapy to touch
- Slippery to feel
- Water soluble base are called alkalis.
- They turn red litmus to blue
EXAMPLES OF BASES
Detergents, baking Soda, drain cleaner, Antacid base, Soaps, lime Water, Milk of Magnesia
TYPES OF BASES
Weak base:-these naturally produce less hydroxide ions in solution.
Strong base:-these produce more number of hydroxide ions on dissolving in water
| NAME OF BASE | FOUND IN |
| Sodium hydroxide | soaps |
| Magnesium hydroxide | antacids |
| Aluminium hydroxide | deodorants |
| Ammonium hydroxide | Window cleaners |
INDICATORS
It is a special chemical that changes its colour to indicate the presence of a chemical substance. It is used to confirm the presence of an acid, a base or a neutral solution.
NATURAL INDICATORS:
1.LITMUS: It is extracted from lichens. It is available in the form of strips or in solution.
2.TURMERIC : It remains yellow in neutral and acidic solutions but turns red in base.
3.CHINA ROSE: It turns acidic solutions to pink and basic solution to green.
4.RED CABBAGE: It turns acidic solutions to red and basic solution to blue.
OTHER INDICATORS
1.METHYL ORANGE : it gives pinkish red colour with acids and yellow colour with bases. 2.PHENOLPHTHALEIN: It is an acid –base indicator . It is colourless in acidic but turns pink in alkali solutions.
NEUTRALIZATION
It is the reaction between an acid and a base which results in the formation of salt and water.
Acid +Base = Salt + Water .
Example :HCl+NaOH= NaCl+ H2O
NEUTRALISATION IN EVERYDAY LIFE
1.INDIGESTION: Too much acid in stomach causes indigestion. It is neutralized by taking an antacid like milk of magnesia.
2.ANT STING :When an ant bites, it injects formic acid into skin .The effect is neutralised by rubbing moist baking soda .
3. SOIL TREATMENT: when the soil is too acidic ,it is neutralized by treating with quick lime .