The Story of discovering the Electromagnetic Spectrum
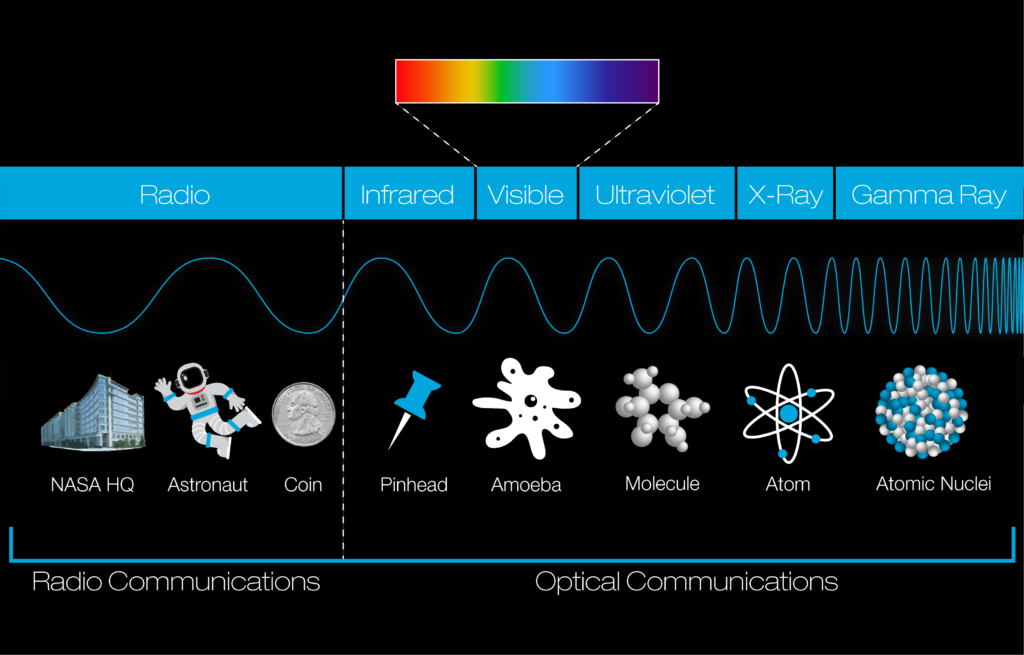
image credit
In physics, light sometimes refers to the electromagnetic radiation of any wavelength, whether visible or not.
There are seven types of electromagnetic radiation in the spectrum, depending on their wavelengths and frequencies. They are :
- Gamma radiation
- X-ray radiation
- Ultraviolet radiation
- Visible light
- Infrared radiation
- Microwave radiation and
- Radio waves
All electromagnetic radiation is light, but we can only see a small portion of this radiation—the one we call visible light.
For most of history, visible light was the only known part of the electromagnetic spectrum.
The ancient Greeks understood that light traveled in straight lines and studied its properties like reflection and refraction.
In 1665, Isaac Newton showed through his experiment that a prism bends visible light and that each color refracts at a slightly different angle depending on its wavelength. Each color in a rainbow corresponds to a different wavelength of the electromagnetic spectrum.

image credit
The first discovery of electromagnetic radiation other than visible light came in 1800 when William Herschel discovered infrared radiation.
An English astronomer William Herschel discovered the planet Uranus in 1781 and was curious about the temperature of different colors. So he placed thermometers in various regions of the rainbow and, as he suspected, different colors registered different temperatures.
Herschel also laid a thermometer outside the spectrum, adjacent to the red, expecting to read no more than room temperature throughout the experiment. But the result was something unexpected. The temperature of his control thermometer rose even higher than in the red.

He theorized that this temperature change was due to calorific rays, a type of light ray that was not visible to see.
Herschel thus accidentally discovered “infra” red light, a brand-new part of the spectrum.
Other investigators immediately took up where Herschel left off. In 1801 the German physicist and pharmacist Johann Wilhelm Ritter found yet another band of invisible light.
Inspired by Herschel’s discovery, he wanted to see if there might be any light just beyond the purple end of the spectrum.
But instead of a thermometer, Ritter placed a little pile of light-sensitive silver chloride in each visible color. He also kept a stack of silver chloride in the dark area next to the violet end of the spectrum. Sure enough, the pile in the unlighted patch darkened more than the pile in the violet patch.
He called them “chemical rays” (invisible light rays that induced chemical reactions). These rays behaved similarly to visible violet light rays but were beyond them in the spectrum. Later these rays were renamed – ultraviolet radiation.
“Ultraviolet” means “beyond violet” (from Latin ultra, “beyond”).
It took a bit longer for scientists to discover the higher-energy (shorter wavelength) light in the electromagnetic spectrum.
Electromagnetic radiation was first linked to electromagnetism in 1845 when Michael Faraday noticed that the polarization of light traveling through a transparent material responded to a magnetic field (Faraday effect).
Around 1860 James Maxwell developed four partial differential equations for the electromagnetic field. Two of these equations predicted the possibility and behavior of waves in the electromagnetic field.
Analyzing the speed of these theoretical waves, Maxwell realized that they must travel at the speed of light, as was known at that time. This startling coincidence in value led Maxwell to conclude – light itself is a type of electromagnetic wave.
Maxwell’s equations predicted an infinite number of frequencies of electromagnetic waves, all traveling at the speed of light. This prediction was the first indication of the existence of the entire electromagnetic spectrum.
In 1887 Heinrich Hertz proved the existence of the waves predicted by Maxwell by producing radio waves in his laboratory.
German physicist Heinrich Hertz had shown that the only real difference among the various kinds of light is the frequency of the waves in each band. Thus he had been credited for recognizing that there is such a thing as an electromagnetic spectrum. In his honor, the unit of frequency has been named the hertz. (frequency – waves per second for anything that vibrates, including sound).
In 1886 in an attempt to prove Maxwell’s equations and detect such low-frequency electromagnetic radiation, the physicist Heinrich Hertz built an apparatus to generate and identify what are now called radio waves.

Image credit
Hertz found the waves and was able to infer (by measuring their wavelength and multiplying it by their frequency) that they traveled at the speed of light.
Hertz also demonstrated that the new radiation could be reflected and refracted by various dielectric media in the same manner as light. In a later experiment, he similarly produced and measured the properties of microwaves.
These new types of waves paved the way for inventions such as the wireless telegraph and the radio.
X-rays were first observed and documented in 1895 by Wilhelm Conrad Röntgen, a German scientist who found them quite by accident when experimenting with vacuum tubes.
A week after he first observed them, he took an X-ray photograph of his wife’s hand, which revealed her wedding ring and her bones. The rays could travel through parts of the human body but were reflected and stopped by denser matter such as bones.
The photo electrified the general public and aroused great scientific interest in the new form of radiation. Röntgen called it “X” to indicate it was an unknown type of radiation. The name stuck, although many of his colleagues suggested calling them Röntgen rays.

While Röntgen first observed the effects of X-rays in 1895, it wasn’t until 1912 that scientists were able to conclude that they were, indeed, another form of light.”
The last segment of the electromagnetic spectrum was filled in – with the discovery of gamma rays. In 1900 Paul Villard was studying the radiation from radium. Then he identified a new type of radiation that he first thought consisted of particles similar to known alpha and beta particles.
Ernest Rutherford is the one who named these radiations gamma rays in 1903 when he realized that they were fundamentally different from charged alpha and beta particles.
However, in 1910, British physicist William Henry Bragg demonstrated that gamma rays are electromagnetic radiation, not particles.
Like X-rays, the exact nature of gamma-rays took a little while for scientists to work out.
And in 1914, Ernest Rutherford and Edward Andrade measured their wavelengths and found that gamma rays were similar to X-rays but with shorter wavelengths and higher frequencies.
In 1914, Rutherford observed that gamma rays reflect in the layers of a crystal. It was clear that they were similar to X-rays. It means they were just another form of light but with much shorter wavelengths.
The electromagnetic spectrum covers electromagnetic waves with frequencies ranging from below 1 hertz to above 1025 hertz, corresponding to wavelengths from thousands of kilometers down to a fraction of the size of an atomic nucleus.
Filling out the entire electromagnetic spectrum, in order of low-energy and low-frequency to high-energy and high-frequency, we have radio waves, microwaves, infrared, ROYGBIV, ultraviolet, X-rays, and gamma rays.
Modern civilization has deftly exploited each of these bands for countless household and industrial applications, making them familiar to us all.